Sepehr Salem Ghahfarokhi
XMorph: Explainable Brain Tumor Analysis Via LLM-Assisted Hybrid Deep Intelligence
Feb 24, 2026Abstract:Deep learning has significantly advanced automated brain tumor diagnosis, yet clinical adoption remains limited by interpretability and computational constraints. Conventional models often act as opaque ''black boxes'' and fail to quantify the complex, irregular tumor boundaries that characterize malignant growth. To address these challenges, we present XMorph, an explainable and computationally efficient framework for fine-grained classification of three prominent brain tumor types: glioma, meningioma, and pituitary tumors. We propose an Information-Weighted Boundary Normalization (IWBN) mechanism that emphasizes diagnostically relevant boundary regions alongside nonlinear chaotic and clinically validated features, enabling a richer morphological representation of tumor growth. A dual-channel explainable AI module combines GradCAM++ visual cues with LLM-generated textual rationales, translating model reasoning into clinically interpretable insights. The proposed framework achieves a classification accuracy of 96.0%, demonstrating that explainability and high performance can co-exist in AI-based medical imaging systems. The source code and materials for XMorph are all publicly available at: https://github.com/ALSER-Lab/XMorph.
Deep learning for automated detection of breast cancer in deep ultraviolet fluorescence images with diffusion probabilistic model
Jul 01, 2024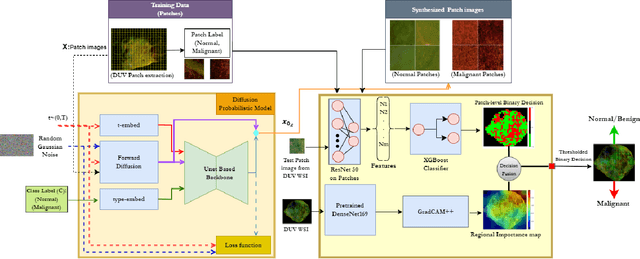
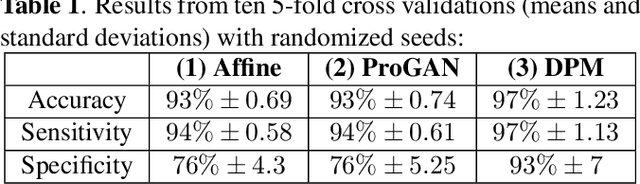

Abstract:Data limitation is a significant challenge in applying deep learning to medical images. Recently, the diffusion probabilistic model (DPM) has shown the potential to generate high-quality images by converting Gaussian random noise into realistic images. In this paper, we apply the DPM to augment the deep ultraviolet fluorescence (DUV) image dataset with an aim to improve breast cancer classification for intraoperative margin assessment. For classification, we divide the whole surface DUV image into small patches and extract convolutional features for each patch by utilizing the pre-trained ResNet. Then, we feed them into an XGBoost classifier for patch-level decisions and then fuse them with a regional importance map computed by Grad-CAM++ for whole surface-level prediction. Our experimental results show that augmenting the training dataset with the DPM significantly improves breast cancer detection performance in DUV images, increasing accuracy from 93% to 97%, compared to using Affine transformations and ProGAN.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge