Navid Ibtehaj Nizam
Enhancing Fluorescence Lifetime Parameter Estimation Accuracy with Differential Transformer Based Deep Learning Model Incorporating Pixelwise Instrument Response Function
Nov 25, 2024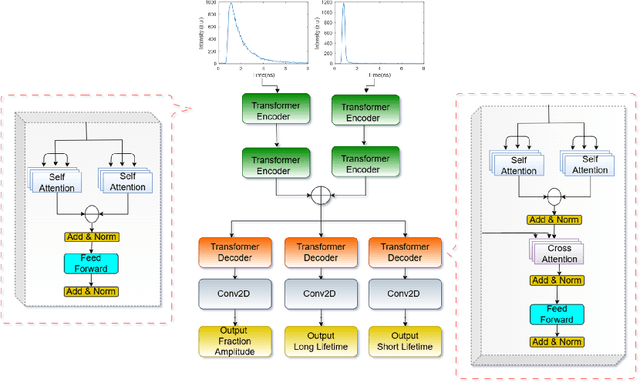
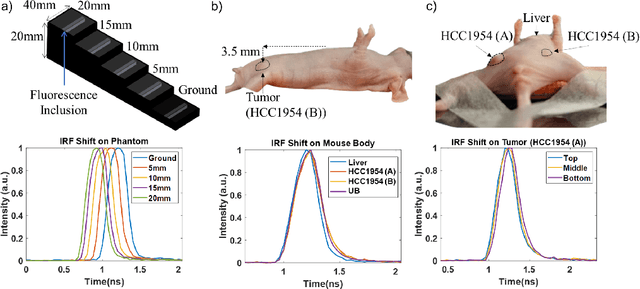
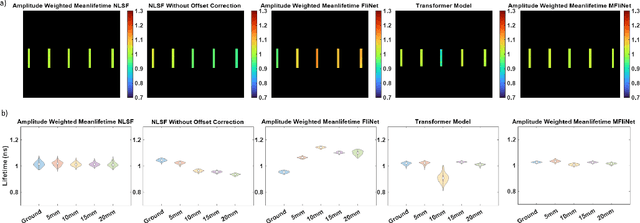
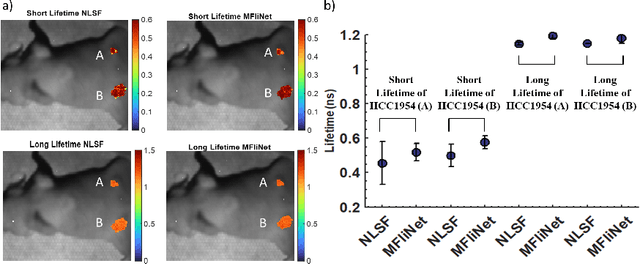
Abstract:Fluorescence lifetime imaging (FLI) is an important molecular imaging modality that can provide unique information for biomedical applications. FLI is based on acquiring and processing photon time of arrival histograms. The shape and temporal offset of these histograms depends on many factors, such as the instrument response function (IRF), optical properties, and the topographic profile of the sample. Several inverse solver analytical methods have been developed to compute the underlying fluorescence lifetime parameters, but most of them are computationally expensive and time-consuming. Thus, deep learning (DL) algorithms have progressively replaced computation methods in fluorescence lifetime parameter estimation. Often, DL models are trained with simple datasets either generated through simulation or a simple experiment where the fluorophore surface profile is mostly flat; therefore, DL models often do not perform well on samples with complex surface profiles such as ex-vivo organs or in-vivo whole intact animals. Herein, we introduce a new DL architecture using state-of-the-art Differential Transformer encoder-decoder architecture, MFliNet (Macroscopic FLI Network), that takes an additional input of IRF together with TPSF, addressing discrepancies in the photon time-of-arrival distribution. We demonstrate the model's performance through carefully designed, complex tissue-mimicking phantoms and preclinical in-vivo cancer xenograft experiments.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge