Leland S. Hu
Mathematical NeuroOncology Lab, Precision Neurotherapeutics Innovation Program, Department of Neurological Surgery, Mayo Clinic, Phoenix, AZ, USA, Department of Diagnostic Imaging and Interventional Radiology, Mayo Clinic, Phoenix, AZ, USA
Knowledge-Informed Machine Learning for Cancer Diagnosis and Prognosis: A review
Jan 12, 2024



Abstract:Cancer remains one of the most challenging diseases to treat in the medical field. Machine learning has enabled in-depth analysis of rich multi-omics profiles and medical imaging for cancer diagnosis and prognosis. Despite these advancements, machine learning models face challenges stemming from limited labeled sample sizes, the intricate interplay of high-dimensionality data types, the inherent heterogeneity observed among patients and within tumors, and concerns about interpretability and consistency with existing biomedical knowledge. One approach to surmount these challenges is to integrate biomedical knowledge into data-driven models, which has proven potential to improve the accuracy, robustness, and interpretability of model results. Here, we review the state-of-the-art machine learning studies that adopted the fusion of biomedical knowledge and data, termed knowledge-informed machine learning, for cancer diagnosis and prognosis. Emphasizing the properties inherent in four primary data types including clinical, imaging, molecular, and treatment data, we highlight modeling considerations relevant to these contexts. We provide an overview of diverse forms of knowledge representation and current strategies of knowledge integration into machine learning pipelines with concrete examples. We conclude the review article by discussing future directions to advance cancer research through knowledge-informed machine learning.
Quantifying intra-tumoral genetic heterogeneity of glioblastoma toward precision medicine using MRI and a data-inclusive machine learning algorithm
Dec 30, 2023Abstract:Glioblastoma (GBM) is one of the most aggressive and lethal human cancers. Intra-tumoral genetic heterogeneity poses a significant challenge for treatment. Biopsy is invasive, which motivates the development of non-invasive, MRI-based machine learning (ML) models to quantify intra-tumoral genetic heterogeneity for each patient. This capability holds great promise for enabling better therapeutic selection to improve patient outcomes. We proposed a novel Weakly Supervised Ordinal Support Vector Machine (WSO-SVM) to predict regional genetic alteration status within each GBM tumor using MRI. WSO-SVM was applied to a unique dataset of 318 image-localized biopsies with spatially matched multiparametric MRI from 74 GBM patients. The model was trained to predict the regional genetic alteration of three GBM driver genes (EGFR, PDGFRA, and PTEN) based on features extracted from the corresponding region of five MRI contrast images. For comparison, a variety of existing ML algorithms were also applied. The classification accuracy of each gene was compared between the different algorithms. The SHapley Additive exPlanations (SHAP) method was further applied to compute contribution scores of different contrast images. Finally, the trained WSO-SVM was used to generate prediction maps within the tumoral area of each patient to help visualize the intra-tumoral genetic heterogeneity. This study demonstrated the feasibility of using MRI and WSO-SVM to enable non-invasive prediction of intra-tumoral regional genetic alteration for each GBM patient, which can inform future adaptive therapies for individualized oncology.
Robust Automatic Whole Brain Extraction on Magnetic Resonance Imaging of Brain Tumor Patients using Dense-Vnet
Jun 04, 2020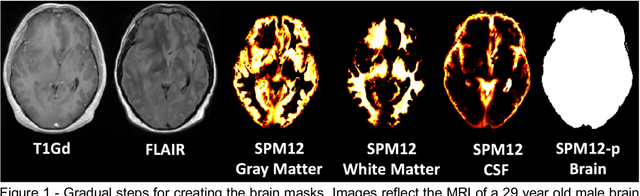
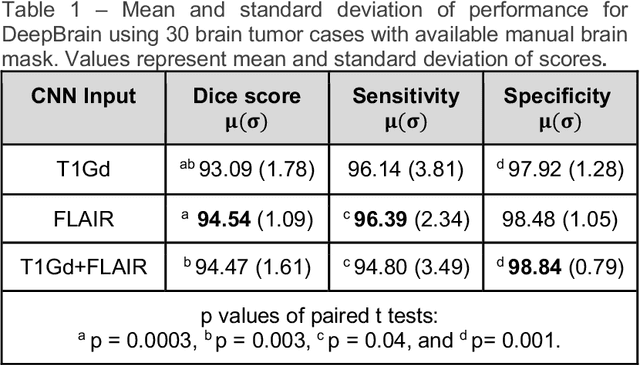
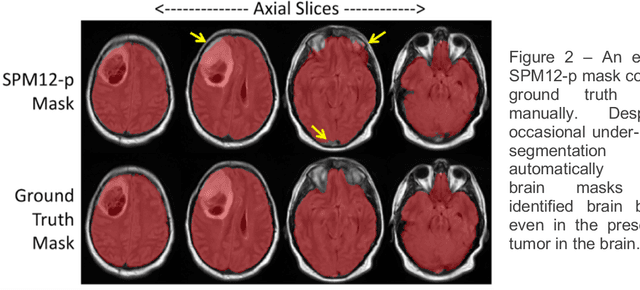
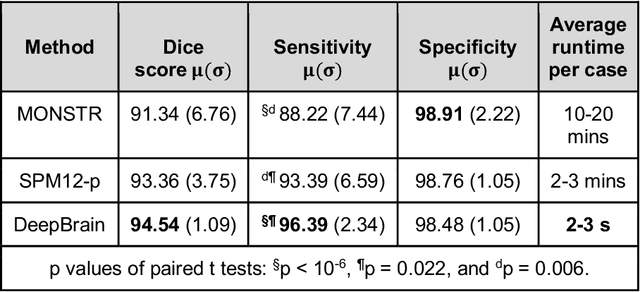
Abstract:Whole brain extraction, also known as skull stripping, is a process in neuroimaging in which non-brain tissue such as skull, eyeballs, skin, etc. are removed from neuroimages. Skull striping is a preliminary step in presurgical planning, cortical reconstruction, and automatic tumor segmentation. Despite a plethora of skull stripping approaches in the literature, few are sufficiently accurate for processing pathology-presenting MRIs, especially MRIs with brain tumors. In this work we propose a deep learning approach for skull striping common MRI sequences in oncology such as T1-weighted with gadolinium contrast (T1Gd) and T2-weighted fluid attenuated inversion recovery (FLAIR) in patients with brain tumors. We automatically created gray matter, white matter, and CSF probability masks using SPM12 software and merged the masks into one for a final whole-brain mask for model training. Dice agreement, sensitivity, and specificity of the model (referred herein as DeepBrain) was tested against manual brain masks. To assess data efficiency, we retrained our models using progressively fewer training data examples and calculated average dice scores on the test set for the models trained in each round. Further, we tested our model against MRI of healthy brains from the LBP40A dataset. Overall, DeepBrain yielded an average dice score of 94.5%, sensitivity of 96.4%, and specificity of 98.5% on brain tumor data. For healthy brains, model performance improved to a dice score of 96.2%, sensitivity of 96.6% and specificity of 99.2%. The data efficiency experiment showed that, for this specific task, comparable levels of accuracy could have been achieved with as few as 50 training samples. In conclusion, this study demonstrated that a deep learning model trained on minimally processed automatically-generated labels can generate more accurate brain masks on MRI of brain tumor patients within seconds.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge