Jon Heiselman
Deformable Registration Framework for Augmented Reality-based Surgical Guidance in Head and Neck Tumor Resection
Mar 11, 2025Abstract:Head and neck squamous cell carcinoma (HNSCC) has one of the highest rates of recurrence cases among solid malignancies. Recurrence rates can be reduced by improving positive margins localization. Frozen section analysis (FSA) of resected specimens is the gold standard for intraoperative margin assessment. However, because of the complex 3D anatomy and the significant shrinkage of resected specimens, accurate margin relocation from specimen back onto the resection site based on FSA results remains challenging. We propose a novel deformable registration framework that uses both the pre-resection upper surface and the post-resection site of the specimen to incorporate thickness information into the registration process. The proposed method significantly improves target registration error (TRE), demonstrating enhanced adaptability to thicker specimens. In tongue specimens, the proposed framework improved TRE by up to 33% as compared to prior deformable registration. Notably, tongue specimens exhibit complex 3D anatomies and hold the highest clinical significance compared to other head and neck specimens from the buccal and skin. We analyzed distinct deformation behaviors in different specimens, highlighting the need for tailored deformation strategies. To further aid intraoperative visualization, we also integrated this framework with an augmented reality-based auto-alignment system. The combined system can accurately and automatically overlay the deformed 3D specimen mesh with positive margin annotation onto the resection site. With a pilot study of the AR guided framework involving two surgeons, the integrated system improved the surgeons' average target relocation error from 9.8 cm to 4.8 cm.
LIBR+: Improving Intraoperative Liver Registration by Learning the Residual of Biomechanics-Based Deformable Registration
Mar 11, 2024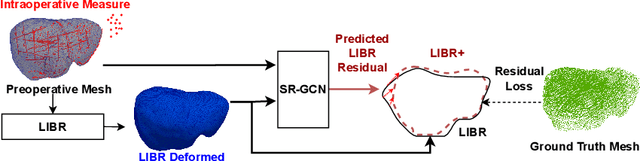

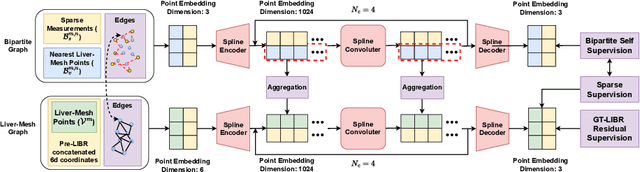

Abstract:The surgical environment imposes unique challenges to the intraoperative registration of organ shapes to their preoperatively-imaged geometry. Biomechanical model-based registration remains popular, while deep learning solutions remain limited due to the sparsity and variability of intraoperative measurements and the limited ground-truth deformation of an organ that can be obtained during the surgery. In this paper, we propose a novel \textit{hybrid} registration approach that leverage a linearized iterative boundary reconstruction (LIBR) method based on linear elastic biomechanics, and use deep neural networks to learn its residual to the ground-truth deformation (LIBR+). We further formulate a dual-branch spline-residual graph convolutional neural network (SR-GCN) to assimilate information from sparse and variable intraoperative measurements and effectively propagate it through the geometry of the 3D organ. Experiments on a large intraoperative liver registration dataset demonstrated the consistent improvements achieved by LIBR+ in comparison to existing rigid, biomechnical model-based non-rigid, and deep-learning based non-rigid approaches to intraoperative liver registration.
Comparing Regularized Kelvinlet Functions and the Finite Element Method for Registration of Medical Images to Sparse Organ Data
Jun 08, 2023



Abstract:Image-guided surgery collocates patient-specific data with the physical environment to facilitate surgical decision making in real-time. Unfortunately, these guidance systems commonly become compromised by intraoperative soft-tissue deformations. Nonrigid image-to-physical registration methods have been proposed to compensate for these deformations, but intraoperative clinical utility requires compatibility of these techniques with data sparsity and temporal constraints in the operating room. While linear elastic finite element models are effective in sparse data scenarios, the computation time for finite element simulation remains a limitation to widespread deployment. This paper proposes a registration algorithm that uses regularized Kelvinlets, which are analytical solutions to linear elasticity in an infinite domain, to overcome these barriers. This algorithm is demonstrated and compared to finite element-based registration on two datasets: a phantom dataset representing liver deformations and an in vivo dataset representing breast deformations. The regularized Kelvinlets algorithm resulted in a significant reduction in computation time compared to the finite element method. Accuracy as evaluated by target registration error was comparable between both methods. Average target registration errors were 4.6 +/- 1.0 and 3.2 +/- 0.8 mm on the liver dataset and 5.4 +/- 1.4 and 6.4 +/- 1.5 mm on the breast dataset for the regularized Kelvinlets and finite element method models, respectively. This work demonstrates the generalizability of using a regularized Kelvinlets registration algorithm on multiple soft tissue elastic organs. This method may improve and accelerate registration for image-guided surgery applications, and it shows the potential of using regularized Kelvinlets solutions on medical imaging data.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge