Dakang Zhang
Predicting the Stereoselectivity of Chemical Transformations by Machine Learning
Oct 12, 2021
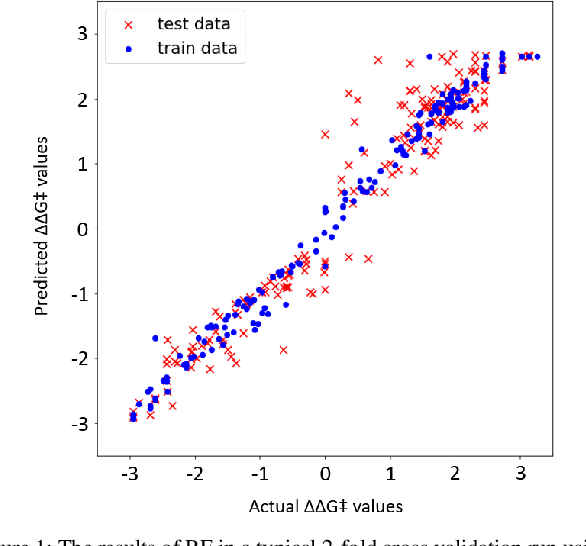
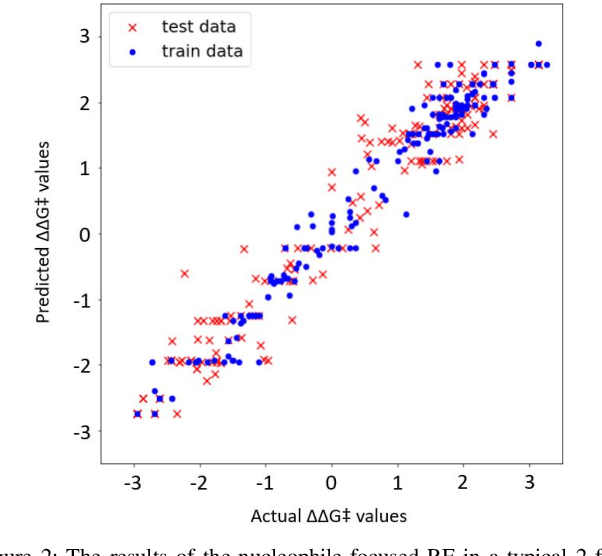
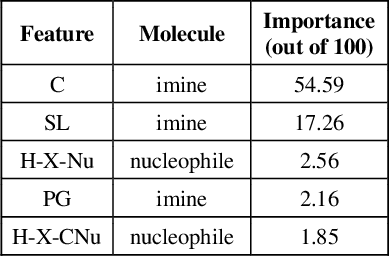
Abstract:Stereoselective reactions (both chemical and enzymatic reactions) have been essential for origin of life, evolution, human biology and medicine. Since late 1960s, there have been numerous successes in the exciting new frontier of asymmetric catalysis. However, most industrial and academic asymmetric catalysis nowadays do follow the trial-and-error model, since the energetic difference for success or failure in asymmetric catalysis is incredibly small. Our current understanding about stereoselective reactions is mostly qualitative that stereoselectivity arises from differences in steric effects and electronic effects in multiple competing mechanistic pathways. Quantitatively understanding and modulating the stereoselectivity of for a given chemical reaction still remains extremely difficult. As a proof of principle, we herein present a novel machine learning technique, which combines a LASSO model and two Random Forest model via two Gaussian Mixture models, for quantitatively predicting stereoselectivity of chemical reactions. Compared to the recent ground-breaking approach [1], our approach is able to capture interactions between features and exploit complex data distributions, which are important for predicting stereoselectivity. Experimental results on a recently published dataset demonstrate that our approach significantly outperform [1]. The insight obtained from our results provide a solid foundation for further exploration of other synthetically valuable yet mechanistically intriguing stereoselective reactions.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge