Andrew Bate
Bayesian dynamic borrowing considering semantic similarity between outcomes for disproportionality analysis in FAERS
Apr 16, 2025Abstract:We present a Bayesian dynamic borrowing (BDB) approach to enhance the quantitative identification of adverse events (AEs) in spontaneous reporting systems (SRSs). The method embeds a robust meta-analytic predictive (MAP) prior within a Bayesian hierarchical model and incorporates semantic similarity measures (SSMs) to enable weighted information sharing from MedDRA Preferred Terms (PTs) that are clinical similar to the target PT. This continuous similarity-based borrowing addresses limitation of rigid hierarchical grouping in current disproportionality analysis (DPA). Using data from the FDA Adverse Event Reporting System (FAERS) between 2015 and 2019, we evalute this approach - termed IC SSM - against standard Information Component (IC) analysis and IC with borrowing at the MedDRA high-level group term (HLGT) level. A novel references set (PVLens), derived from FDA product label updates, enabled prospective evaluation of method performance in identifying AEs prior to official labeling. The IC SSM approach demonstrated improved sensitivity compared to both traditional IC and HLGT-based borrowing, with minor trade-offs in F1 scores and Youden's index. IC SSM consistently identified more true positives and detected signals over 5 months sooner than traditional IC. Despite a marginally lower aggregate Youden's index, IC SSM showed higher performance in the early post-marketing period, providing more stable and relevant estimates than HLGT-based borrowing and traditional IC. These findings support the use of SSM-informed Bayesian borrowing as a scalable and context-aware enhancement to traditional DPA methods. Future research should validate this approach across other datasets and explore additional similarity metrics and Bayesian inference strategies using case-level data.
PVLens: Enhancing Pharmacovigilance Through Automated Label Extraction
Mar 27, 2025Abstract:Reliable drug safety reference databases are essential for pharmacovigilance, yet existing resources like SIDER are outdated and static. We introduce PVLens, an automated system that extracts labeled safety information from FDA Structured Product Labels (SPLs) and maps terms to MedDRA. PVLens integrates automation with expert oversight through a web-based review tool. In validation against 97 drug labels, PVLens achieved an F1 score of 0.882, with high recall (0.983) and moderate precision (0.799). By offering a scalable, more accurate and continuously updated alternative to SIDER, PVLens enhances real-time pharamcovigilance with improved accuracy and contemporaneous insights.
Ontology-based Semantic Similarity Measures for Clustering Medical Concepts in Drug Safety
Mar 26, 2025Abstract:Semantic similarity measures (SSMs) are widely used in biomedical research but remain underutilized in pharmacovigilance. This study evaluates six ontology-based SSMs for clustering MedDRA Preferred Terms (PTs) in drug safety data. Using the Unified Medical Language System (UMLS), we assess each method's ability to group PTs around medically meaningful centroids. A high-throughput framework was developed with a Java API and Python and R interfaces support large-scale similarity computations. Results show that while path-based methods perform moderately with F1 scores of 0.36 for WUPALMER and 0.28 for LCH, intrinsic information content (IC)-based measures, especially INTRINSIC-LIN and SOKAL, consistently yield better clustering accuracy (F1 score of 0.403). Validated against expert review and standard MedDRA queries (SMQs), our findings highlight the promise of IC-based SSMs in enhancing pharmacovigilance workflows by improving early signal detection and reducing manual review.
Bridging the Gap in Drug Safety Data Analysis: Large Language Models for SQL Query Generation
Jun 15, 2024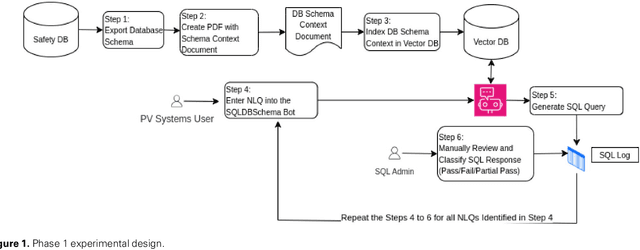
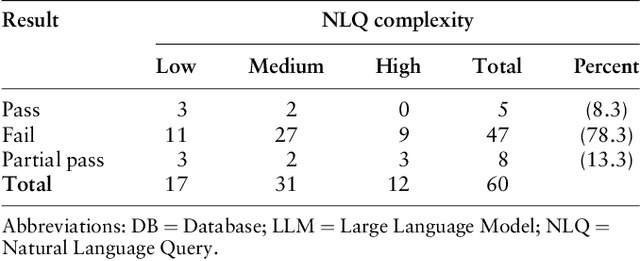


Abstract:Pharmacovigilance (PV) is essential for drug safety, primarily focusing on adverse event monitoring. Traditionally, accessing safety data required database expertise, limiting broader use. This paper introduces a novel application of Large Language Models (LLMs) to democratize database access for non-technical users. Utilizing OpenAI's GPT-4, we developed a chatbot that generates structured query language (SQL) queries from natural language, bridging the gap between domain knowledge and technical requirements. The proposed application aims for more inclusive and efficient data access, enhancing decision making in drug safety. By providing LLMs with plain language summaries of expert knowledge, our approach significantly improves query accuracy over methods relying solely on database schemas. The application of LLMs in this context not only optimizes PV data analysis, ensuring timely and precise drug safety reporting -- a crucial component in adverse drug reaction monitoring -- but also promotes safer pharmacological practices and informed decision making across various data intensive fields.
Extending Consequence-Based Reasoning to SRIQ
Feb 23, 2016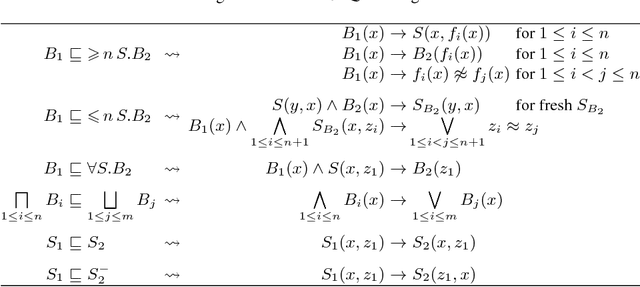

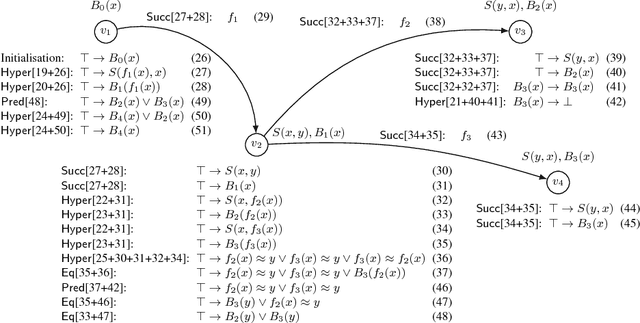

Abstract:Consequence-based calculi are a family of reasoning algorithms for description logics (DLs), and they combine hypertableau and resolution in a way that often achieves excellent performance in practice. Up to now, however, they were proposed for either Horn DLs (which do not support disjunction), or for DLs without counting quantifiers. In this paper we present a novel consequence-based calculus for SRIQ---a rich DL that supports both features. This extension is non-trivial since the intermediate consequences that need to be derived during reasoning cannot be captured using DLs themselves. The results of our preliminary performance evaluation suggest the feasibility of our approach in practice.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge