Adrian Gericke
SurGrID: Controllable Surgical Simulation via Scene Graph to Image Diffusion
Feb 11, 2025Abstract:Surgical simulation offers a promising addition to conventional surgical training. However, available simulation tools lack photorealism and rely on hardcoded behaviour. Denoising Diffusion Models are a promising alternative for high-fidelity image synthesis, but existing state-of-the-art conditioning methods fall short in providing precise control or interactivity over the generated scenes. We introduce SurGrID, a Scene Graph to Image Diffusion Model, allowing for controllable surgical scene synthesis by leveraging Scene Graphs. These graphs encode a surgical scene's components' spatial and semantic information, which are then translated into an intermediate representation using our novel pre-training step that explicitly captures local and global information. Our proposed method improves the fidelity of generated images and their coherence with the graph input over the state-of-the-art. Further, we demonstrate the simulation's realism and controllability in a user assessment study involving clinical experts. Scene Graphs can be effectively used for precise and interactive conditioning of Denoising Diffusion Models for simulating surgical scenes, enabling high fidelity and interactive control over the generated content.
Synthesising Rare Cataract Surgery Samples with Guided Diffusion Models
Aug 03, 2023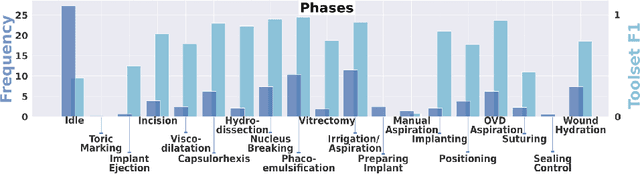
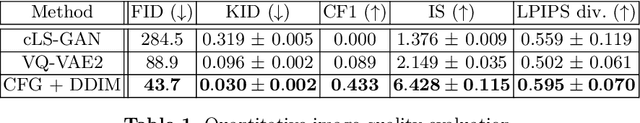
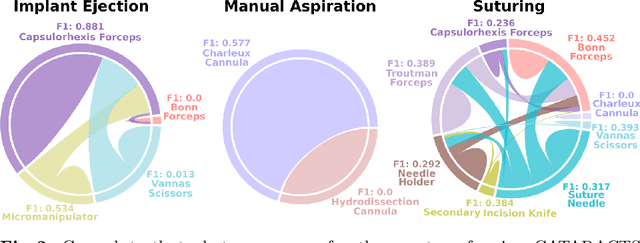
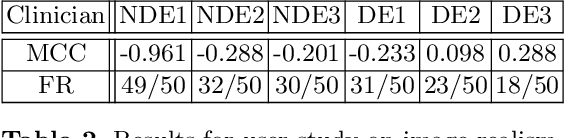
Abstract:Cataract surgery is a frequently performed procedure that demands automation and advanced assistance systems. However, gathering and annotating data for training such systems is resource intensive. The publicly available data also comprises severe imbalances inherent to the surgical process. Motivated by this, we analyse cataract surgery video data for the worst-performing phases of a pre-trained downstream tool classifier. The analysis demonstrates that imbalances deteriorate the classifier's performance on underrepresented cases. To address this challenge, we utilise a conditional generative model based on Denoising Diffusion Implicit Models (DDIM) and Classifier-Free Guidance (CFG). Our model can synthesise diverse, high-quality examples based on complex multi-class multi-label conditions, such as surgical phases and combinations of surgical tools. We affirm that the synthesised samples display tools that the classifier recognises. These samples are hard to differentiate from real images, even for clinical experts with more than five years of experience. Further, our synthetically extended data can improve the data sparsity problem for the downstream task of tool classification. The evaluations demonstrate that the model can generate valuable unseen examples, allowing the tool classifier to improve by up to 10% for rare cases. Overall, our approach can facilitate the development of automated assistance systems for cataract surgery by providing a reliable source of realistic synthetic data, which we make available for everyone.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge