Tobias Geyer
AI-Driven Decision Support in Oncology: Evaluating Data Readiness for Skin Cancer Treatment
Mar 12, 2025



Abstract:This research focuses on evaluating and enhancing data readiness for the development of an Artificial Intelligence (AI)-based Clinical Decision Support System (CDSS) in the context of skin cancer treatment. The study, conducted at the Skin Tumor Center of the University Hospital M\"unster, delves into the essential role of data quality, availability, and extractability in implementing effective AI applications in oncology. By employing a multifaceted methodology, including literature review, data readiness assessment, and expert workshops, the study addresses the challenges of integrating AI into clinical decision-making. The research identifies crucial data points for skin cancer treatment decisions, evaluates their presence and quality in various information systems, and highlights the difficulties in extracting information from unstructured data. The findings underline the significance of high-quality, accessible data for the success of AI-driven CDSS in medical settings, particularly in the complex field of oncology.
Declarative Guideline Conformance Checking of Clinical Treatments: A Case Study
Sep 20, 2022
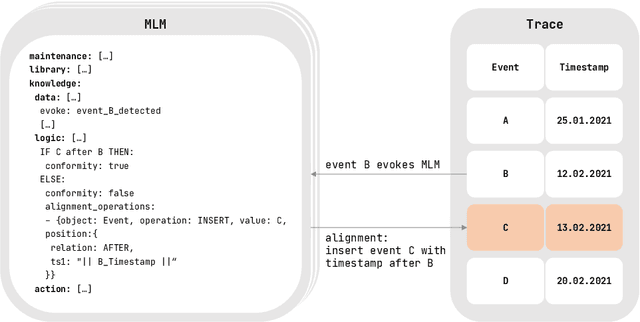
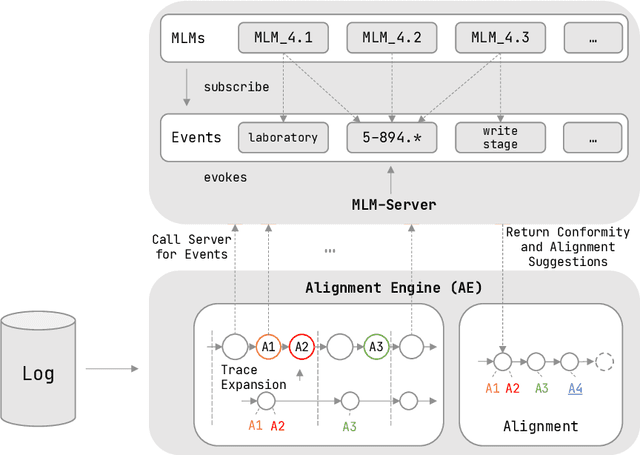
Abstract:Conformance checking is a process mining technique that allows verifying the conformance of process instances to a given model. Thus, this technique is predestined to be used in the medical context for the comparison of treatment cases with clinical guidelines. However, medical processes are highly variable, highly dynamic, and complex. This makes the use of imperative conformance checking approaches in the medical domain difficult. Studies show that declarative approaches can better address these characteristics. However, none of the approaches has yet gained practical acceptance. Another challenge are alignments, which usually do not add any value from a medical point of view. For this reason, we investigate in a case study the usability of the HL7 standard Arden Syntax for declarative, rule-based conformance checking and the use of manually modeled alignments. Using the approach, it was possible to check the conformance of treatment cases and create medically meaningful alignments for large parts of a medical guideline.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge