Joe de Keizer
G-computation for increasing performances of clinical trials with individual randomization and binary response
Nov 15, 2024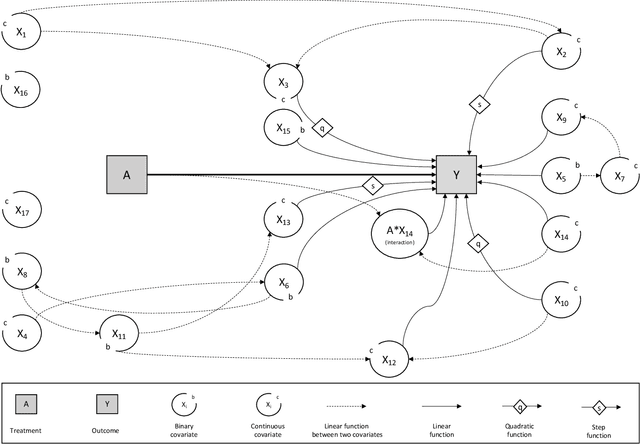
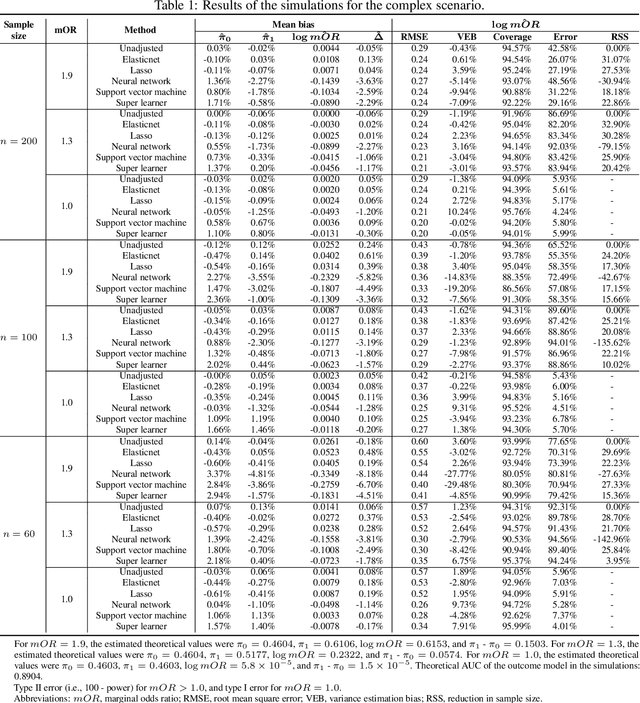
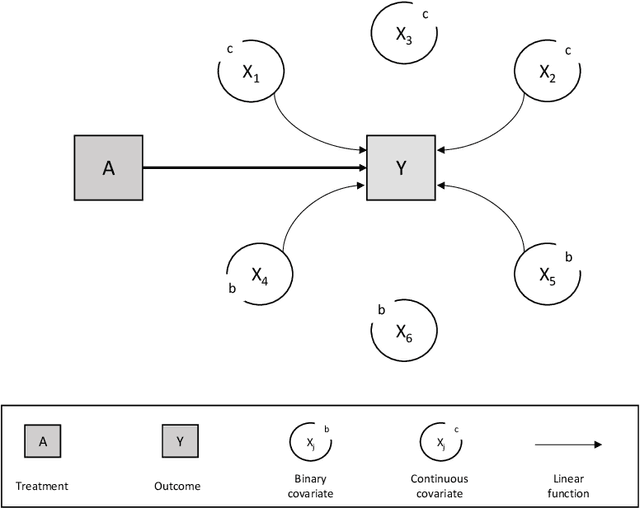
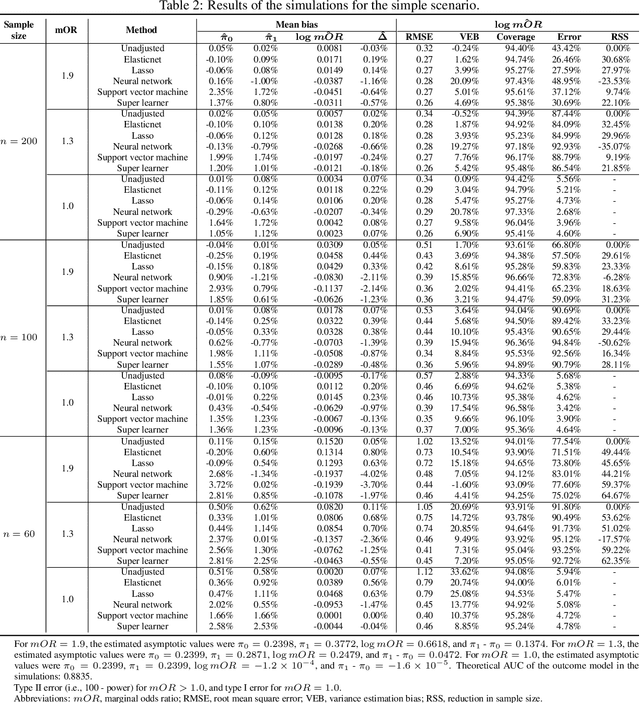
Abstract:In a clinical trial, the random allocation aims to balance prognostic factors between arms, preventing true confounders. However, residual differences due to chance may introduce near-confounders. Adjusting on prognostic factors is therefore recommended, especially because the related increase of the power. In this paper, we hypothesized that G-computation associated with machine learning could be a suitable method for randomized clinical trials even with small sample sizes. It allows for flexible estimation of the outcome model, even when the covariates' relationships with outcomes are complex. Through simulations, penalized regressions (Lasso, Elasticnet) and algorithm-based methods (neural network, support vector machine, super learner) were compared. Penalized regressions reduced variance but may introduce a slight increase in bias. The associated reductions in sample size ranged from 17\% to 54\%. In contrast, algorithm-based methods, while effective for larger and more complex data structures, underestimated the standard deviation, especially with small sample sizes. In conclusion, G-computation with penalized models, particularly Elasticnet with splines when appropriate, represents a relevant approach for increasing the power of RCTs and accounting for potential near-confounders.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge