Henry Westmacott
Unsupervised Learning of Object-Centric Embeddings for Cell Instance Segmentation in Microscopy Images
Oct 12, 2023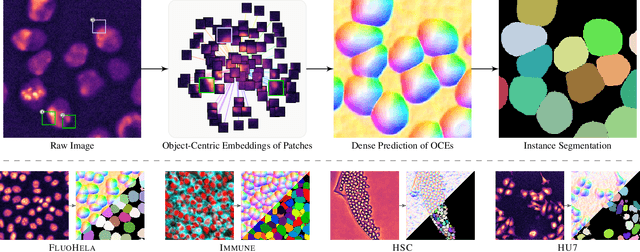
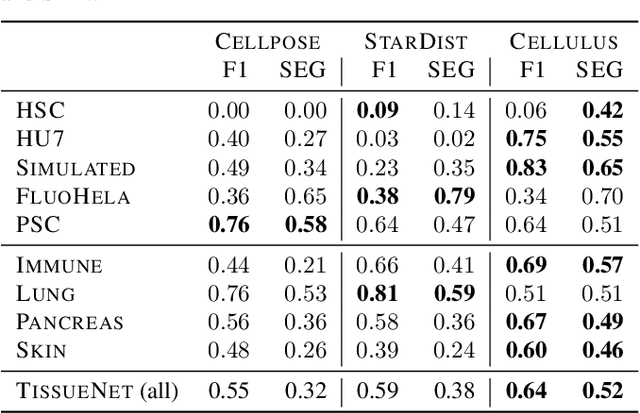
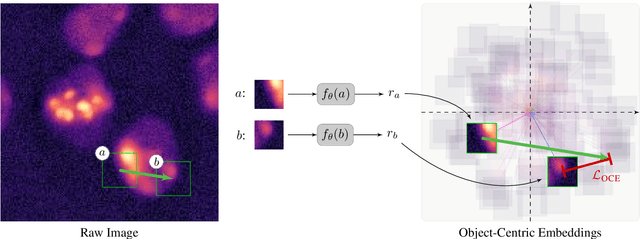
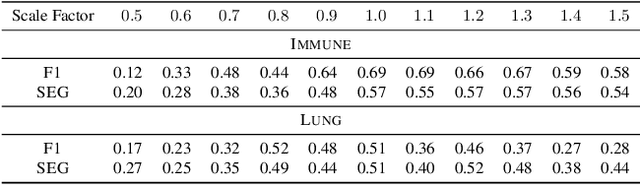
Abstract:Segmentation of objects in microscopy images is required for many biomedical applications. We introduce object-centric embeddings (OCEs), which embed image patches such that the spatial offsets between patches cropped from the same object are preserved. Those learnt embeddings can be used to delineate individual objects and thus obtain instance segmentations. Here, we show theoretically that, under assumptions commonly found in microscopy images, OCEs can be learnt through a self-supervised task that predicts the spatial offset between image patches. Together, this forms an unsupervised cell instance segmentation method which we evaluate on nine diverse large-scale microscopy datasets. Segmentations obtained with our method lead to substantially improved results, compared to state-of-the-art baselines on six out of nine datasets, and perform on par on the remaining three datasets. If ground-truth annotations are available, our method serves as an excellent starting point for supervised training, reducing the required amount of ground-truth needed by one order of magnitude, thus substantially increasing the practical applicability of our method. Source code is available at https://github.com/funkelab/cellulus.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge