Gokhan Bora Esmer
Super-Resolution for Interferometric Imaging: Model Comparisons and Performance Analysis
Feb 21, 2025Abstract:This study investigates the application of Super-Resolution techniques in holographic microscopy to enhance quantitative phase imaging. An off-axis Mach-Zehnder interferometric setup was employed to capture interferograms. The study evaluates two Super-Resolution models, RCAN and Real-ESRGAN, for their effectiveness in reconstructing high-resolution interferograms from a microparticle-based dataset. The models were assessed using two primary approaches: image-based analysis for structural detail enhancement and morphological evaluation for maintaining sample integrity and phase map accuracy. The results demonstrate that RCAN achieves superior numerical precision, making it ideal for applications requiring highly accurate phase map reconstruction, while Real-ESRGAN enhances visual quality and structural coherence, making it suitable for visualization-focused applications. This study highlights the potential of Super-Resolution models in overcoming diffraction-imposed resolution limitations in holographic microscopy, opening the way for improved imaging techniques in biomedical diagnostics, materials science, and other high-precision fields.
Holographic Cell Stiffness Mapping Using Acoustic Stimulation
Feb 15, 2021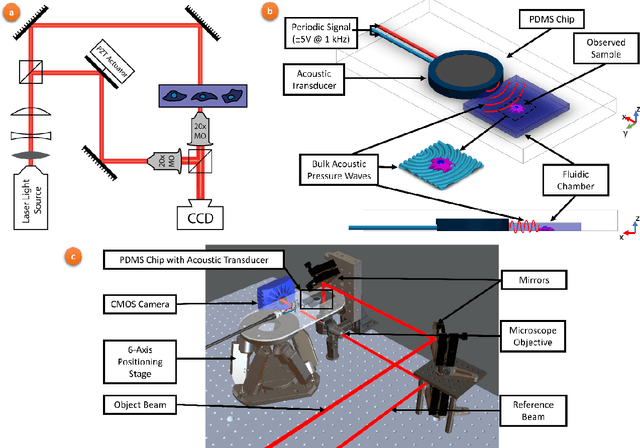
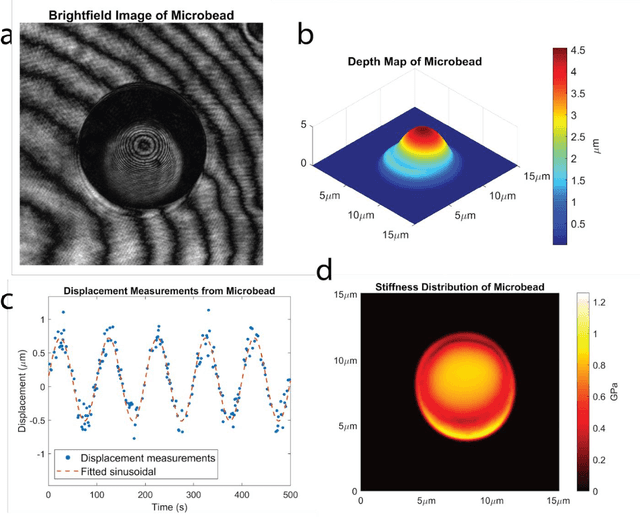
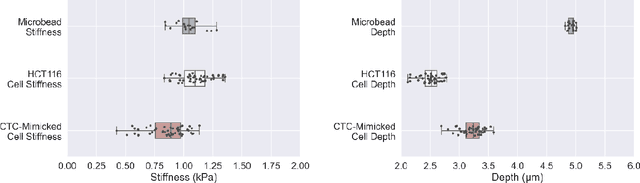
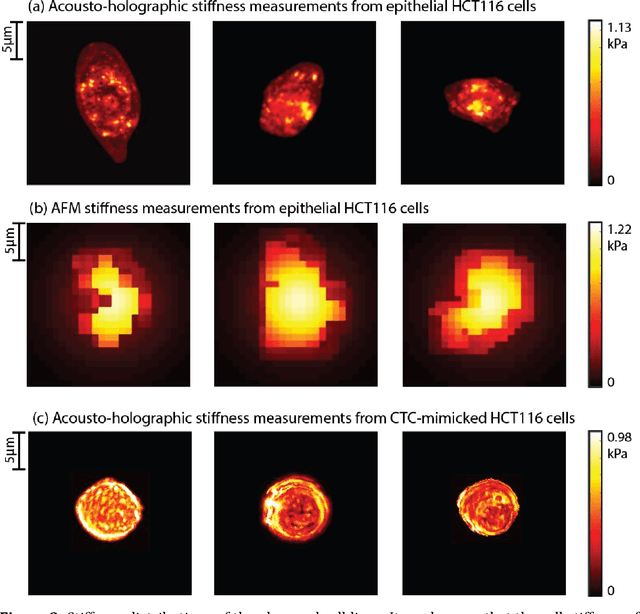
Abstract:Accurate assessment of stiffness distribution is essential due to the critical role of single cell mechanobiology in the regulation of many vital cellular processes such as proliferation, adhesion, migration, and motility. Cell stiffness is one of the fundamental mechanical properties of the cell and is greatly affected by the intracellular tensional forces, cytoskeletal prestress, and cytoskeleton structure. Herein, we propose a novel holographic single-cell stiffness measurement technique that can obtain the stiffness distribution over a cell membrane at high resolution and in real-time. The proposed imaging method coupled with acoustic signals allows us to assess the cell stiffness distribution with a low error margin and label-free manner. We demonstrate the proposed technique on HCT116 (Human Colorectal Carcinoma) cells and CTC-mimicked HCT116 cells by induction with transforming growth factor-beta (TGF-\b{eta}). Validation studies of the proposed approach were carried out on certified polystyrene microbeads with known stiffness levels. Its performance was evaluated in comparison with the AFM results obtained for the relevant cells. When the experimental results were examined, the proposed methodology shows utmost performance over average cell stiffness values for HCT116, and CTC-mimicked HCT116 cells were found as 1.08 kPa, and 0.88 kPa, respectively. The results confirm that CTC-mimicked HCT116 cells lose their adhesion ability to enter the vascular circulation and metastasize. They also exhibit a softer stiffness profile compared to adherent forms of the cancer cells. Hence, the proposed technique is a significant, reliable, and faster alternative for in-vitro cell stiffness characterization tools. It can be utilized for various applications where single-cell analysis is required, such as disease modeling, drug testing, diagnostics, and many more.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge