Jutika Borah
FALCON: Few-Shot Adversarial Learning for Cross-Domain Medical Image Segmentation
Jan 04, 2026Abstract:Precise delineation of anatomical and pathological structures within 3D medical volumes is crucial for accurate diagnosis, effective surgical planning, and longitudinal disease monitoring. Despite advancements in AI, clinically viable segmentation is often hindered by the scarcity of 3D annotations, patient-specific variability, data privacy concerns, and substantial computational overhead. In this work, we propose FALCON, a cross-domain few-shot segmentation framework that achieves high-precision 3D volume segmentation by processing data as 2D slices. The framework is first meta-trained on natural images to learn-to-learn generalizable segmentation priors, then transferred to the medical domain via adversarial fine-tuning and boundary-aware learning. Task-aware inference, conditioned on support cues, allows FALCON to adapt dynamically to patient-specific anatomical variations across slices. Experiments on four benchmarks demonstrate that FALCON consistently achieves the lowest Hausdorff Distance scores, indicating superior boundary accuracy while maintaining a Dice Similarity Coefficient comparable to the state-of-the-art models. Notably, these results are achieved with significantly less labeled data, no data augmentation, and substantially lower computational overhead.
DCAT: Dual Cross-Attention Fusion for Disease Classification in Radiological Images with Uncertainty Estimation
Mar 14, 2025Abstract:Accurate and reliable image classification is crucial in radiology, where diagnostic decisions significantly impact patient outcomes. Conventional deep learning models tend to produce overconfident predictions despite underlying uncertainties, potentially leading to misdiagnoses. Attention mechanisms have emerged as powerful tools in deep learning, enabling models to focus on relevant parts of the input data. Combined with feature fusion, they can be effective in addressing uncertainty challenges. Cross-attention has become increasingly important in medical image analysis for capturing dependencies across features and modalities. This paper proposes a novel dual cross-attention fusion model for medical image analysis by addressing key challenges in feature integration and interpretability. Our approach introduces a bidirectional cross-attention mechanism with refined channel and spatial attention that dynamically fuses feature maps from EfficientNetB4 and ResNet34 leveraging multi-network contextual dependencies. The refined features through channel and spatial attention highlights discriminative patterns crucial for accurate classification. The proposed model achieved AUC of 99.75%, 100%, 99.93% and 98.69% and AUPR of 99.81%, 100%, 99.97%, and 96.36% on Covid-19, Tuberculosis, Pneumonia Chest X-ray images and Retinal OCT images respectively. The entropy values and several high uncertain samples give an interpretable visualization from the model enhancing transparency. By combining multi-scale feature extraction, bidirectional attention and uncertainty estimation, our proposed model strongly impacts medical image analysis.
Disease Classification and Impact of Pretrained Deep Convolution Neural Networks on Diverse Medical Imaging Datasets across Imaging Modalities
Sep 02, 2024


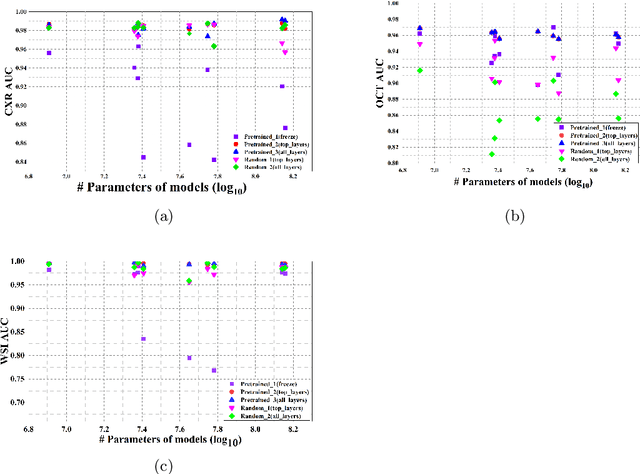
Abstract:Imaging techniques such as Chest X-rays, whole slide images, and optical coherence tomography serve as the initial screening and detection for a wide variety of medical pulmonary and ophthalmic conditions respectively. This paper investigates the intricacies of using pretrained deep convolutional neural networks with transfer learning across diverse medical imaging datasets with varying modalities for binary and multiclass classification. We conducted a comprehensive performance analysis with ten network architectures and model families each with pretraining and random initialization. Our finding showed that the use of pretrained models as fixed feature extractors yields poor performance irrespective of the datasets. Contrary, histopathology microscopy whole slide images have better performance. It is also found that deeper and more complex architectures did not necessarily result in the best performance. This observation implies that the improvements in ImageNet are not parallel to the medical imaging tasks. Within a medical domain, the performance of the network architectures varies within model families with shifts in datasets. This indicates that the performance of models within a specific modality may not be conclusive for another modality within the same domain. This study provides a deeper understanding of the applications of deep learning techniques in medical imaging and highlights the impact of pretrained networks across different medical imaging datasets under five different experimental settings.
Predictive uncertainty estimation in deep learning for lung carcinoma classification in digital pathology under real dataset shifts
Aug 15, 2024Abstract:Deep learning has shown tremendous progress in a wide range of digital pathology and medical image classification tasks. Its integration into safe clinical decision-making support requires robust and reliable models. However, real-world data comes with diversities that often lie outside the intended source distribution. Moreover, when test samples are dramatically different, clinical decision-making is greatly affected. Quantifying predictive uncertainty in models is crucial for well-calibrated predictions and determining when (or not) to trust a model. Unfortunately, many works have overlooked the importance of predictive uncertainty estimation. This paper evaluates whether predictive uncertainty estimation adds robustness to deep learning-based diagnostic decision-making systems. We investigate the effect of various carcinoma distribution shift scenarios on predictive performance and calibration. We first systematically investigate three popular methods for improving predictive uncertainty: Monte Carlo dropout, deep ensemble, and few-shot learning on lung adenocarcinoma classification as a primary disease in whole slide images. Secondly, we compare the effectiveness of the methods in terms of performance and calibration under clinically relevant distribution shifts such as in-distribution shifts comprising primary disease sub-types and other characterization analysis data; out-of-distribution shifts comprising well-differentiated cases, different organ origin, and imaging modality shifts. While studies on uncertainty estimation exist, to our best knowledge, no rigorous large-scale benchmark compares predictive uncertainty estimation including these dataset shifts for lung carcinoma classification.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge