Granger causal inference on DAGs identifies genomic loci regulating transcription
Paper and Code
Oct 18, 2022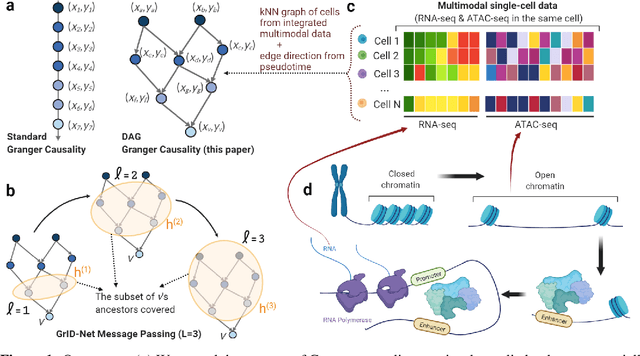
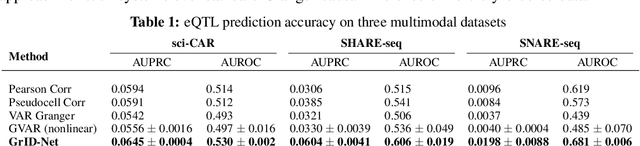
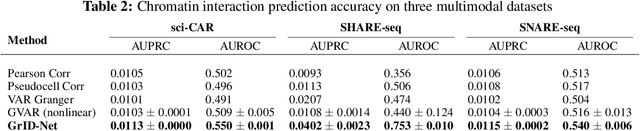
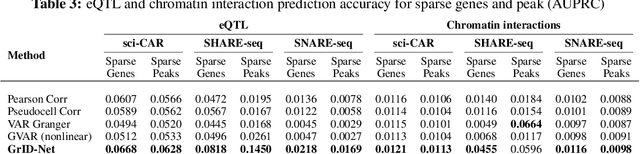
When a dynamical system can be modeled as a sequence of observations, Granger causality is a powerful approach for detecting predictive interactions between its variables. However, traditional Granger causal inference has limited utility in domains where the dynamics need to be represented as directed acyclic graphs (DAGs) rather than as a linear sequence, such as with cell differentiation trajectories. Here, we present GrID-Net, a framework based on graph neural networks with lagged message passing for Granger causal inference on DAG-structured systems. Our motivating application is the analysis of single-cell multimodal data to identify genomic loci that mediate the regulation of specific genes. To our knowledge, GrID-Net is the first single-cell analysis tool that accounts for the temporal lag between a genomic locus becoming accessible and its downstream effect on a target gene's expression. We applied GrID-Net on multimodal single-cell assays that profile chromatin accessibility (ATAC-seq) and gene expression (RNA-seq) in the same cell and show that it dramatically outperforms existing methods for inferring regulatory locus-gene links, achieving up to 71% greater agreement with independent population genetics-based estimates. By extending Granger causality to DAG-structured dynamical systems, our work unlocks new domains for causal analyses and, more specifically, opens a path towards elucidating gene regulatory interactions relevant to cellular differentiation and complex human diseases at unprecedented scale and resolution.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge